Comprehensive
Home Care
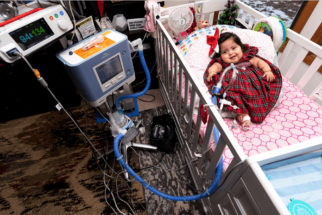
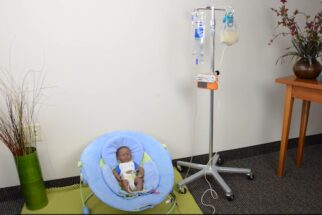
Pediatric Home Service (PHS) wants families to be together at home. That’s why we have a comprehensive care model that covers all of your needs. Our services include Respiratory & Equipment, Enteral Nutrition, Infusion Nursing & Pharmacy, Home Care/Private Duty Nursing, and more. Our goal is to help kids with medical complexities live as fully as possible – at home, with their families.